
Ultimate Guide to Hydrogen Fuel Cell Materials
Share
Hydrogen fuel cells generate electricity by combining hydrogen and oxygen, producing only water and heat as byproducts. These systems are efficient and offer a cleaner energy alternative to fossil fuels. The key to their performance lies in the materials used for components like electrodes, membranes, and catalysts. Here's what you need to know:
- Core Components: Fuel cells include an anode, cathode, proton exchange membrane (PEM), and catalyst layers.
- Materials Matter: Platinum is a common catalyst, but alternatives like nickel-based and single-atom catalysts are reducing costs. Modern PEMs, such as PFSA polymers, improve performance and durability.
- Efficiency and Cost: Advances in thinner membranes, non-platinum catalysts, and lightweight materials are improving efficiency while lowering costs.
- Applications: Fuel cells power vehicles, stationary systems, and portable devices, with each application requiring specific material choices.
Hydrogen fuel cells continue to evolve, addressing cost and durability challenges while expanding their use across industries.
Tech Talk - Catalyst & Catalyst Support for Fuel Cells - Fuel Cell Tech Explained - Hyfindr Arlene
Main Components and Materials in Hydrogen Fuel Cells
Hydrogen fuel cells rely on four key components: the anode, the cathode, the proton exchange membrane (PEM), and the catalyst layers. The materials used for these components play a major role in determining the fuel cell's overall performance and lifespan.
Anode and Cathode Materials
In Proton Exchange Membrane Fuel Cells (PEMFCs), the anode and cathode are where the critical electrochemical reactions occur. These electrodes are typically made from carbon-based substrates like carbon paper, carbon cloth, or graphite. These materials are chosen for their excellent conductivity and resistance to corrosion. More advanced options, such as carbon nanotubes and graphene, are being explored to reduce internal resistance and increase the active surface area, which can further enhance efficiency and durability.
The choice of catalyst depends on the type of fuel cell. In PEMFCs, platinum (Pt) is commonly used at the anode for hydrogen oxidation, while platinum alloys (e.g., Pt–Co or Pt–Ni) are used at the cathode to facilitate oxygen reduction. In contrast, Solid Oxide Fuel Cells (SOFCs) operate at higher temperatures, allowing the use of nickel-based cermets (like Ni–YSZ) for the anode and perovskite oxides, such as LSCF or LSM, for the cathode. For instance, the 2020 Toyota Mirai employs platinum loadings of about 0.05 mg/cm² at the anode and 0.315 mg/cm² at the cathode, reflecting the slower oxygen reaction kinetics at the cathode.
Both the anode and cathode are designed to be porous. This structure maximizes the reaction area at the triple-phase boundary, where gas, liquid, and solid phases meet. This design choice underscores the importance of the proton exchange membrane in driving fuel cell performance.
Proton Exchange Membranes (PEMs)
The proton exchange membrane serves two critical purposes: it allows protons to move from the anode to the cathode while forcing electrons to travel through an external circuit, generating electricity. Most PEMs are made from perfluorosulfonic acid (PFSA) polymers. These materials feature a hydrophobic PTFE backbone for mechanical and chemical stability, along with hydrophilic side chains ending in sulfonic acid groups that facilitate proton transport.
"The PTFE main chain is hydrophobic and determines the mechanical and chemical stability of the proton exchange membrane. The sulfonic acid end groups are the primary active components for proton conductivity."
When hydrated, the membrane forms tiny water channels that allow protons to move through two mechanisms: the vehicular method (as hydronium ions, H₃O⁺) and the Grotthuss mechanism (proton hopping). Modern PFSA membranes are now often thinner than 10 µm, a significant improvement over older versions like Nafion N117, which was 180 µm thick. This reduction in thickness lowers resistance and boosts performance. Short side chain (SSC) membranes, such as Aquivion from Solvay-Solexis, provide higher ionic conductivity and better performance under low-humidity conditions compared to traditional long side chain membranes.
Standard PFSA membranes operate best between 80–90°C, while phosphoric acid-doped PBI membranes can handle much higher temperatures, ranging from 100°C to 250°C. Cost-wise, the membrane accounts for about 17% of the total stack cost at low production volumes (1,000 systems/year), dropping to around 9% when production scales to 50,000 systems/year.
Catalyst Layers and Nanoparticles
Catalysts are essential for speeding up the reactions at both electrodes. At the anode, they break hydrogen molecules into protons and electrons, while at the cathode, they help reduce oxygen to form water. Platinum remains the go-to catalyst due to its optimal oxygen binding energy, but its high cost - around $30,000 per kilogram - has led to efforts to reduce its usage.
Nanoparticles have become critical in catalyst layers, as their high surface-area-to-volume ratio increases the number of active sites at the triple-phase boundary. For oxygen reduction reactions, the ideal size for platinum nanoparticles is about 3 nanometers, balancing activity and stability. Advances in this technology have significantly lowered platinum usage, with anode loadings dropping from 1 mg/cm² in 2000 to approximately 0.025 mg/cm² by 2021. Additionally, cobalt-platinum (Co–Pt) alloy nanocatalysts have achieved power outputs of 5.9 kW per gram of platinum, nearly doubling current commercial standards.
"We have developed a platform technology to produce a complicated form of alloy nanocatalysts through a simple and scalable method, and finally achieved an enhanced fuel cell power performance with less amount of platinum used."
- Prof. Hyeon Taeghwan, Director of the Center for Nanoparticle Research, Institute for Basic Science
Typically, nanoparticles are supported on high-surface-area carbon materials, such as Vulcan XC 72, which provide excellent electrical conductivity and structural stability. Despite advances, the catalyst layer remains one of the most expensive components of a PEM fuel cell stack, accounting for nearly 42% of the total cost in high-volume production (around 500,000 units/year).
Recent Developments in Fuel Cell Materials
Recent progress in fuel cell technology has centered on cutting costs while improving performance. The focus has been on three main areas: identifying alternatives to pricey platinum catalysts, enhancing membrane durability, and designing lighter, more corrosion-resistant components.
Non-Platinum Catalyst Options
Nickel-based catalysts are emerging as a cost-effective alternative for hydrogen oxidation in alkaline settings. Their abundance and platinum-like properties make them a compelling choice. In September 2025, a team from Huazhong University of Science and Technology mapped out promising options, including MoNi₄ and Ni₃N nanoparticles. These materials demonstrate performance levels nearing that of platinum in alkaline fuel cells.
Single-atom catalysts (SACs), especially iron–nitrogen–carbon (FeNC) materials, are another exciting development. They maximize metal efficiency while addressing cost and scarcity issues. Pajarito Powder LLC has been producing FeNC catalysts at an industrial scale since 2014. Their method uses a sacrificial silica support to create porous carbon networks with active FeN₄ sites. As Florian D. Speck from the Helmholtz-Institute Erlangen-Nürnberg notes:
Single-atom catalysts (SACs) hold great promise for maximized metal utilization, exceptional tunability of the catalytic site, and selectivity.
The growing adoption of alkaline anion-exchange membrane fuel cells (AEMFCs) is also noteworthy. These systems allow for the use of cheaper catalysts, such as nickel and iron, unlike traditional proton-exchange membrane cells that rely on platinum. However, hydrogen oxidation in alkaline environments is significantly slower than in acidic ones. Advances in alloy engineering, like NiCu alloys and ternary Ni–Mo–Nb metallic glasses, are addressing this by optimizing hydrogen binding energy and speeding up catalytic reactions. These innovations in catalysts are also enhancing membrane performance.
Improved Membrane Technologies
Membrane technology has moved beyond traditional polymer designs. In February 2026, a Stanford University team led by Yi Cui and Jinwei Xu introduced a hydrogen-molecule-mediated proton exchange membrane. This design transports hydrogen molecules through a gas chamber and electrons through a metal, bypassing conventional proton conduction. It achieves nearly 100% proton selectivity and an area-specific resistance of 0.15 Ω·cm² at pH 0, solving the usual trade-offs between selectivity and conductivity.
High-temperature PEMs (160–180°C), made with phosphoric acid-doped PBI-based membranes, also offer significant advantages. They can handle carbon monoxide concentrations up to 3 vol%, unlike their low-temperature counterparts, which are highly sensitive to such impurities. Additionally, Pt–Ni and Pt–Co alloy catalysts on reduced graphene oxide (rGO) supports have shown better resistance to ripening in high-temperature phosphoric acid environments compared to traditional carbon-supported catalysts.
Another breakthrough involves ionomer-free gas diffusion cathodes created using atomic layer deposition (ALD). These electrodes, with platinum loadings as low as 1.08 μg/cm² - a dramatic 99.5% reduction compared to commercial standards - have maintained stability at 1 A/cm² for over 200 hours. By precisely placing nanoparticles at the membrane–microporous layer interface, these ultra-thin catalyst layers improve both catalyst efficiency and mass transport. These advancements in membranes are complemented by improvements in structural materials.
Lightweight and Corrosion-Resistant Materials
Bipolar plates, which account for 80% of a fuel cell stack's weight and 50–65% of its volume, are now a prime target for weight reduction. Stainless steel and titanium are replacing graphite due to their strength, reduced thickness, and compatibility with 3D printing.
The acidic environment of PEM fuel cells can corrode metals, releasing ions that damage catalysts and disrupt ion transport. To combat this, a molybdenum gradient enrichment layer on 2205 duplex stainless steel has been developed. This layer forms a protective n–p type bilayer semiconductor, significantly reducing membrane degradation.
Metal nitride coatings like CrN and TiN are also proving effective, offering a balance of conductivity, corrosion protection, and industrial practicality. Multilayer coating designs, with their dense, defect-blocking structures, can cut corrosion current density by over 80% compared to single-layer coatings. New manufacturing techniques such as selective laser melting (SLM) and advanced 3D printing now allow for intricate, lightweight flow field designs with ultra-thin profiles - often less than 0.1 mm - previously unattainable with traditional methods. These material advances are key to improving the overall performance and longevity of hydrogen fuel cells.
sbb-itb-501186b
Choosing Materials for Optimal Performance
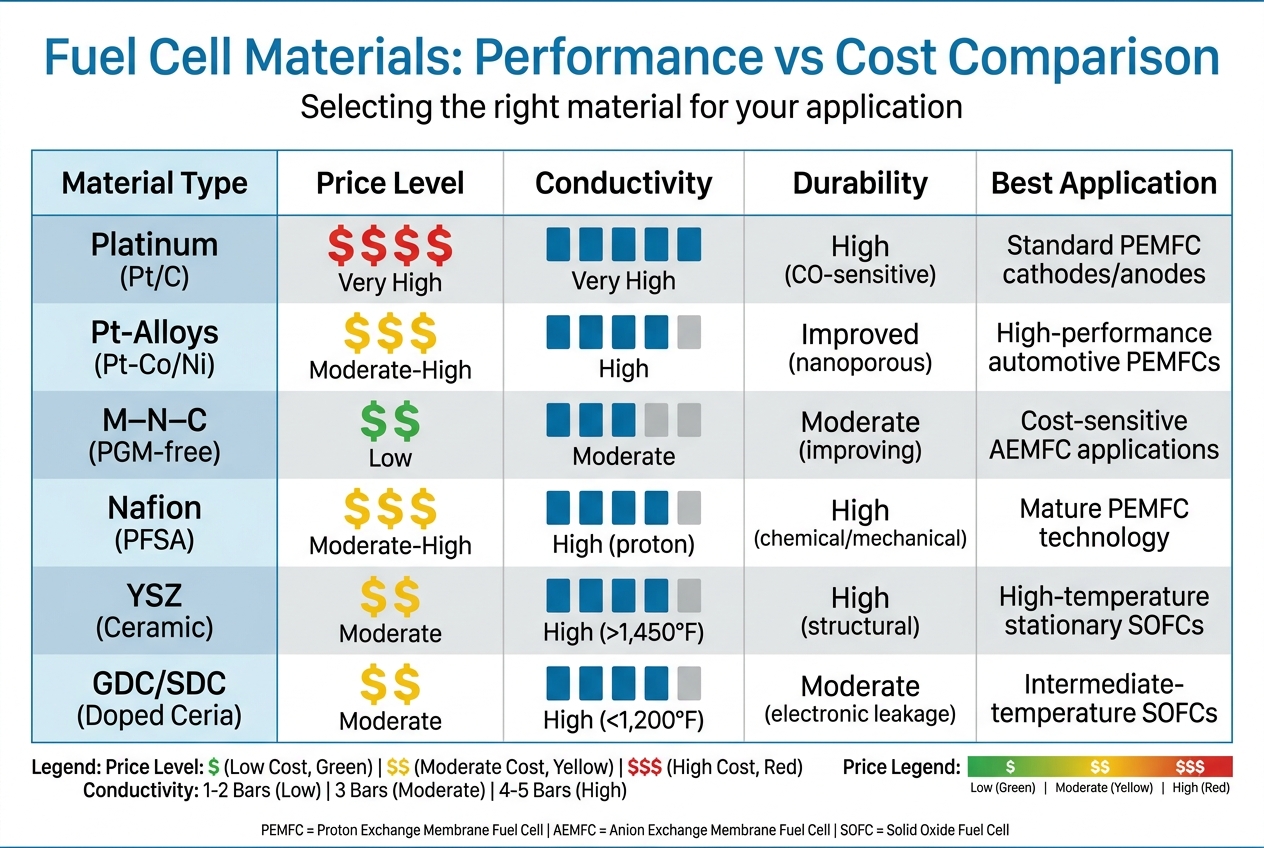
Hydrogen Fuel Cell Materials: Performance vs Cost Comparison
Recent advancements in materials have opened up new possibilities for improving fuel cell efficiency. However, making the right material choices means carefully balancing technical requirements with cost considerations. This balance directly impacts the lifespan, power output, and overall cost of fuel cells.
Material Compatibility Requirements
Fuel cell components operate under extreme conditions. The anode must withstand a reducing environment, while the cathode endures an oxidizing one - all while maintaining structural stability over long periods. Electrolytes play a key role, needing ionic conductivities of at least 1 × 10⁻² S cm⁻¹ at the operating temperature to ensure smooth performance.
Thermal expansion compatibility is another critical factor, especially in high-temperature systems. If the Coefficient of Thermal Expansion (CTE) between components - like the cathode and electrolyte - doesn't match, it can lead to delamination and even complete stack failure during thermal cycling.
Chemical stability is equally vital. For instance, the electrolyte must act as an effective barrier to prevent fuel and oxidant from mixing directly. Any breach would not only lower efficiency but also pose safety concerns.
While technical performance is crucial, cost efficiency remains a major focus when selecting materials.
Balancing Performance and Cost
Platinum-based catalysts dominate the cost of Membrane Electrode Assemblies, accounting for about 91% of the total expense. This makes material selection a high-stakes decision. The table below outlines some common material choices:
| Material Type | Price | Conductivity | Durability | Best Application |
|---|---|---|---|---|
| Platinum (Pt/C) | Very High | Very High | High (CO-sensitive) | Standard PEMFC cathodes/anodes |
| Pt-Alloys (Pt-Co/Ni) | Moderate-High | High | Improved (nanoporous) | High-performance automotive PEMFCs |
| M–N–C (PGM-free) | Low | Moderate | Moderate (improving) | Cost-sensitive AEMFC applications |
| Nafion (PFSA) | Moderate-High | High (proton) | High (chemical/mechanical) | Mature PEMFC technology |
| YSZ (Ceramic) | Moderate | High (>1,450°F) | High (structural) | High-temperature stationary SOFCs |
| GDC/SDC (Doped Ceria) | Moderate | High (<1,200°F) | Moderate (electronic leakage) | Intermediate-temperature SOFCs |
To cut costs without sacrificing too much performance, modern PEMFC stacks now use ultra-thin membranes - typically between 8 and 10 µm. These thinner membranes reduce ohmic losses while keeping material expenses in check. For applications where affordability is more important than peak performance, Anion Exchange Membrane Fuel Cells (AEMFCs) are a strong option. These systems use non-precious metal catalysts, with PGM-free cathodes now achieving peak power densities exceeding 1 W cm⁻².
Temperature adjustments also offer cost-saving opportunities. By lowering Solid Oxide Fuel Cell operating temperatures from over 1,470°F to a range of 840–1,290°F, manufacturers can switch to less expensive stainless steel interconnects and simpler sealing materials. This significantly reduces system costs while maintaining solid performance levels.
Where Hydrogen Fuel Cells Are Used
Hydrogen fuel cells power a wide range of applications, from vehicles to industrial backup systems and even portable devices. Their performance and reliability hinge on the materials used, which are carefully selected to meet the unique demands of each application.
Transportation Sector
Proton Exchange Membrane Fuel Cells (PEMFCs) dominate the transportation sector, particularly in fuel cell vehicles, due to their ability to operate at relatively low temperatures (below 100°C or 212°F). This allows for quick startups and responsive power delivery, making them ideal for on-the-go use. South Korea currently leads with 19,270 fuel cell vehicles on the road, followed by the United States with 12,283 vehicles. Globally, passenger cars make up about 82% of fuel cell vehicles, while buses and trucks account for 9.2% and 8.7%, respectively.
To enhance performance, transportation applications rely on lightweight and durable materials. For instance, ultra-thin membranes improve efficiency, while bipolar plates have evolved from graphite to coated metals to better handle vibrations and corrosion. Many fuel cell vehicles now feature hybrid powertrains that pair the fuel cell with a battery. This setup captures energy from regenerative braking and supports peak power demands.
Infrastructure is also expanding rapidly. By 2021, there were 685 hydrogen refueling stations worldwide, enabling broader adoption of fuel cell vehicles. Models like the Toyota Mirai and Hyundai Nexo showcase advancements in fuel cell technology, emphasizing quick startup times and hybrid designs tailored for automotive use.
Stationary Power Systems
Stationary power systems, such as those used for backup power in data centers, microgrids, and industrial facilities, often employ high-temperature fuel cells like Solid Oxide Fuel Cells (SOFCs) and Molten Carbonate Fuel Cells (MCFCs). These systems are built for long-term reliability rather than fast startups.
High-temperature systems require materials that can endure extreme conditions. Ceramic components, for example, must withstand thermal cycling without cracking. Perovskite oxide cathodes, like LSCF and LSM, offer electronic conductivities of around 1,000 S cm⁻¹ while resisting oxidation. Electrolytes need to maintain ionic conductivity of at least 1 × 10⁻² S cm⁻¹ at operating temperatures for smooth operation. Because high temperatures accelerate reaction kinetics, nickel-based catalysts are often used as a cost-effective alternative to platinum. Additionally, anode materials are designed to resist carbon buildup and sulfur poisoning, using solutions like perovskite oxides or metallic alloy catalysts with ceria. These material innovations ensure that stationary systems remain reliable over time.
Portable Power Applications
Portable fuel cells, such as Direct Methanol Fuel Cells (DMFCs) and compact PEMFCs, are tailored for situations where size and energy density are critical. By using liquid fuels, these systems avoid the need for bulky hydrogen storage, making them ideal for military gear, remote sensors, and emergency power supplies.
To maximize efficiency, portable systems incorporate specialized membranes that minimize fuel crossover while balancing cost and performance. Lightweight, high-conductivity carbon-based electrodes are also a key feature. These carefully chosen materials highlight how hydrogen fuel cell technology continues to evolve to meet the needs of portable and compact applications.
Conclusion and Key Takeaways
Hydrogen fuel cell materials have made significant progress in addressing hurdles like cost, durability, and performance. For instance, platinum currently makes up about 91% of the cost of Membrane Electrode Assemblies (MEAs), which has spurred the development of alternative catalysts. Among these, metal–nitrogen–carbon (M–N–C) compounds stand out, achieving power densities greater than 1 W cm⁻². Meanwhile, high-temperature PEM fuel cells with polybenzimidazole (PBI) membranes now handle carbon monoxide concentrations of up to 3%, a stark contrast to the 10–100 ppm tolerance of low-temperature systems. This makes them a practical option for reformed hydrogen sourced from methanol or ammonia.
Recent advancements in catalyst and membrane technology have significantly boosted fuel cell performance under practical conditions. In 2025, researchers at Blue World Technologies showcased Celtec-based MEAs capable of operating for thousands of hours at 320°F (160°C) using reformed methanol. They also introduced carbon-nanocage-supported PtCo catalysts, which demonstrated improved mass activity. Additionally, thinner membranes have been developed to reduce ohmic losses and enhance water management.
A shift toward intermediate-temperature fuel cells, functioning in the 390–750°F (200–400°C) range, is striking a balance between faster reaction rates and material longevity. New SDC@Al₂O₃ core–shell composite electrolytes have achieved ionic conductivity of 0.096 S cm⁻¹ and power densities of 1,190 mW cm⁻² at 1,022°F (550°C). These breakthroughs are helping to create cleaner and more efficient energy systems.
Hydrogen fuel cells hold the potential to cut emissions in heavy transport by up to 67% by 2050. With the PEM fuel cell materials market projected to surpass $2.5 billion by 2036, continued advancements in materials are crucial for making these systems economically viable. Moreover, the integration of digital twins and AI optimization is extending fuel cell lifespans by managing factors like thermal stress and fuel variability.
FAQs
Why is the cathode catalyst the biggest cost driver in PEM fuel cells?
The cathode catalyst stands out as the biggest expense in PEM fuel cells. Why? Because it depends on platinum-based materials, which are both rare and costly. To meet the required catalytic activity, a high amount of platinum is needed, which only adds to the overall expense.
What trade-offs come with using ultra-thin PEM membranes?
Ultra-thin PEM (proton exchange membrane) membranes in hydrogen fuel cells offer the potential to cut costs by as much as 20% while boosting eco-friendly advantages. However, making membranes thinner comes with trade-offs. It can reduce durability, weaken mechanical stability, and lower thermal resistance, leaving the membranes more vulnerable to wear and tear. Additionally, thinner membranes may allow more methanol to pass through, which can negatively affect both performance and lifespan. To overcome these hurdles, precise engineering is crucial to maintain reliability over time.
When should a project choose PEMFCs vs AEMFCs vs SOFCs based on materials?
Proton Exchange Membrane Fuel Cells (PEMFCs) are a great choice for applications requiring quick startups, a compact design, and operation at lower temperatures (below 120°C). These qualities make them particularly well-suited for backup power systems and transportation needs.
On the other hand, Alkaline Exchange Membrane Fuel Cells (AEMFCs) shine in scenarios where reducing dependence on precious metals is a priority. However, they do face hurdles, especially when it comes to membrane conductivity, which can impact their performance.
For high-efficiency applications that require operation at elevated temperatures (above 800°C), Solid Oxide Fuel Cells (SOFCs) are the go-to option. They excel in stationary power generation but are less practical for portable or backup power solutions due to their longer startup times.
