
How PEM Electrolysis Transforms Hydrogen Production
Share
PEM (Proton Exchange Membrane) electrolysis is reshaping how hydrogen is produced, offering a cleaner, more efficient alternative to traditional methods. By splitting water into hydrogen and oxygen using electricity and a polymer membrane, it creates high-purity hydrogen without carbon emissions - especially when powered by renewable energy sources like wind or solar. Here's what you need to know:
- Efficiency: PEM electrolysis achieves up to 80% efficiency, operating at 122°F–176°F and producing hydrogen at pressures of 30–34 bar, reducing the need for additional compression.
- High-Purity Hydrogen: Produces hydrogen with purity levels of 99.9% or higher, suitable for fuel cell vehicles and industrial uses.
- Dynamic Response: Quickly adjusts to power fluctuations, making it ideal for capturing surplus renewable energy.
- Cost Goals: The U.S. Department of Energy aims to reduce hydrogen production costs from $6.40/kg in 2023 to $2.00/kg by 2026 and $1.00/kg by 2031.
PEM technology is compact, flexible, and offers direct high-pressure hydrogen production, but it currently has higher upfront costs due to the use of precious metal catalysts like platinum and iridium. Ongoing research is focused on reducing these costs while scaling up production.
In short, PEM electrolysis is a key player in the transition to cleaner energy systems, enabling hydrogen to serve as a viable, zero-emission fuel.
How A PEM Electrolyser Works (Lesson 2/5) Hunor Kacso | Hydrogen Training Solutions
sbb-itb-501186b
How PEM Electrolysis Works
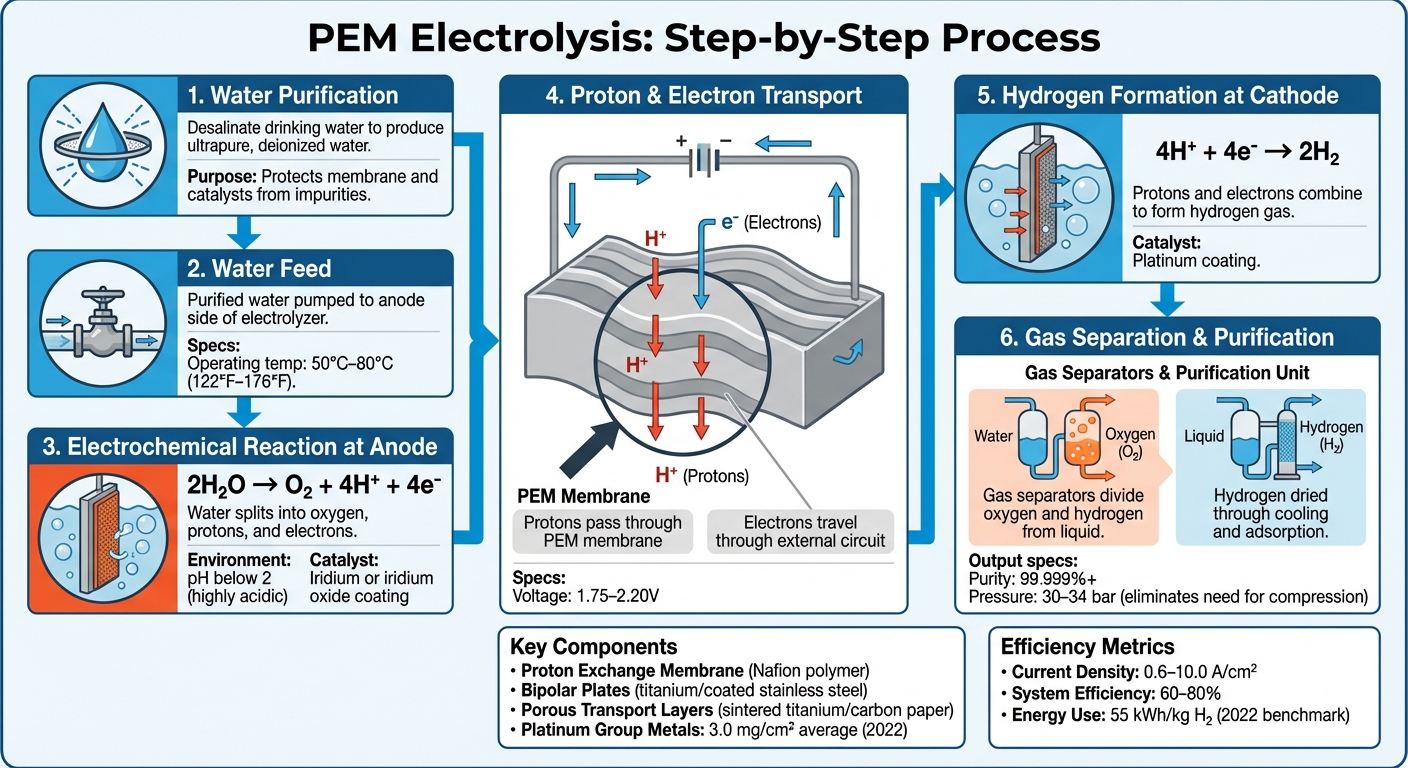
How PEM Electrolysis Works: 6-Step Water-to-Hydrogen Process
The Electrochemical Process
PEM electrolysis operates through a series of electrochemical reactions that occur on either side of a thin polymer membrane. At the anode, water molecules break apart in a process called oxidation. This reaction produces oxygen gas, protons, and electrons, following the equation:
2H₂O → O₂ + 4H⁺ + 4e⁻.
This reaction takes place in a highly acidic environment, with a pH below 2 - comparable to battery acid.
The protons pass through the proton exchange membrane to the cathode, while the electrons are forced to move through an external circuit since the membrane blocks their direct passage. At the cathode, the protons and electrons combine to form hydrogen gas, as described by:
4H⁺ + 4e⁻ → 2H₂.
The system typically operates at temperatures between 50°C–80°C (122°F–176°F) and voltages of 1.75–2.20V, taking into account internal resistances.
Key Components of a PEM Electrolyzer
A PEM electrolyzer is made up of several specialized components working together. At its core is the proton exchange membrane, often made from a solid polymer like Nafion. This membrane conducts protons while preventing gases and electrons from passing through. Despite being only 100–200 micrometers thick, it can handle pressures of up to 34 bar.
The electrodes, coated with precious metals, drive the chemical reactions. The anode typically uses iridium or iridium oxide to facilitate oxygen evolution, while the cathode relies on platinum to produce hydrogen. Bipolar plates, usually made from titanium or coated stainless steel, provide structural support and ensure uniform electrical current distribution. Porous transport layers - such as sintered titanium on the anode side and carbon paper on the cathode - help distribute water evenly and remove the resulting gases efficiently.
As of 2022, PEM electrolyzer stacks contained an average of 3.0 mg/cm² of platinum group metals (PGMs). Efforts are underway, such as the EU's Clean Hydrogen Partnership, to lower PGM usage from 2.5 mg/W to 0.25 mg/W by 2030, aiming to reduce costs.
Steps in PEM Electrolysis
The process begins with desalinating drinking water to produce ultrapure, deionized water. This step is crucial to protect the membrane and catalysts from impurities. The purified water is then pumped into the system and fed to the anode side of the electrolyzer.
When direct current is applied, the electrochemical reactions kick off. At the anode, water splits into protons (which pass through the membrane) and electrons (which travel through an external circuit). The resulting mixtures - oxygen-rich water at the anode and hydrogen-rich water at the cathode - are directed to gas separators to separate the gases from the liquid.
The hydrogen is then purified and dried through cooling and adsorption processes to remove residual moisture, achieving purity levels that often exceed 99.999%. Many systems produce hydrogen at pressures between 30 and 34 bar, reducing or even eliminating the need for additional mechanical compression.
Looking ahead, Bosch plans to launch its Hybrion PEM electrolysis stacks for industrial use in 2025. Each stack, made up of over 100 cells, is designed to produce up to 22.9 kg of hydrogen per hour with a power input of 1.25 megawatts and an output pressure of 34 bar.
These carefully orchestrated steps highlight the precision and efficiency of PEM electrolysis in producing green hydrogen.
Benefits of PEM Electrolysis
What Makes PEM Different?
PEM electrolysis stands out with several advantages that make it a strong contender in modern energy systems. One of its key strengths is its high current density. PEM electrolyzers typically operate in a range of 0.6 to 10.0 A/cm², and some research setups have even achieved peak densities of up to 20 A/cm². This means more hydrogen can be produced using a smaller electrode area.
Another standout feature is the compact design. Thanks to thin solid polymer membranes - measuring just 100–200 micrometers - and high power density, PEM systems require much less space compared to other technologies. These compact units can even fit inside standard shipping containers, making them ideal for industrial environments where space is limited.
PEM electrolyzers also excel in dynamic flexibility, quickly adapting to changes in renewable energy input. This responsiveness ensures efficient operation, even at partial loads.
Additionally, PEM technology produces high-pressure hydrogen directly, typically in the range of 15 to 34 bar (220–500 psi). This can significantly reduce or even eliminate the need for extra mechanical compression equipment. The solid membrane also acts as a built-in gas separator, achieving hydrogen purity levels that often exceed 99.999%.
In terms of efficiency, current PEM systems operate within a range of 60–80%, with benchmarks of 55 kWh/kg H₂ (61% LHV) reported in 2022. The U.S. Department of Energy aims to improve these numbers, targeting 51 kWh/kg H₂ (65% LHV) by 2026 while also working to extend system lifetimes from 40,000 to 80,000 operational hours.
These features make PEM electrolysis a competitive option, especially when compared to alkaline electrolysis.
PEM vs. Alkaline Electrolysis
When comparing PEM and alkaline electrolysis, the differences become clear. Here’s how they stack up:
| Feature | PEM Electrolysis | Alkaline Electrolysis |
|---|---|---|
| Current Density | High (0.6–10.0 A/cm²) | Low (0.2–0.4 A/cm²) |
| Energy Efficiency | 51–55 kWh/kg H₂ | 62–82% efficiency |
| Output Pressure | 15–34 bar | Atmospheric to low pressure |
| Operating Temp. | 68–194°F (20–90°C) | 86–194°F (30–90°C) |
| Renewable Adaptability | High (fast response) | Low (sluggish response) |
| System Footprint | Compact/Small | Large |
In practical use, the differences are even more pronounced. Alkaline systems often face challenges with partial load operation and require warm-up cycles, whereas PEM systems can adjust almost instantly to power fluctuations. This adaptability has earned PEM electrolysis recognition for its ability to handle renewable energy variability. As the Carbon Neutrality Journal puts it:
"hailed as the most desired technology for high purity hydrogen production and self‐consistent with volatility of renewable energies".
However, PEM systems come with higher upfront costs. As of 2022, they were priced at approximately $1,000/kW - more expensive than alkaline systems. But there’s good news: the U.S. Department of Energy has set ambitious goals to bring PEM costs down to $250/kW by 2026 and eventually to $150/kW. These reductions are expected through innovations like decreasing the use of precious metals (from 0.8 g/kW to 0.1 g/kW) and scaling up manufacturing.
For applications requiring integration with renewable energy or high-purity hydrogen at elevated pressures, PEM’s flexibility, compact size, and output quality often make it worth the higher initial investment.
Applications of PEM Electrolysis
Green Hydrogen Production
PEM electrolysis is becoming a cornerstone of industrial-scale green hydrogen production, thanks to its efficiency and ability to respond quickly to changes in energy supply. One of its key roles is storing surplus renewable energy as hydrogen, providing a solution for long-term energy storage.
A notable example is Air Liquide's 200 MW PEM electrolyzer project in Port-Jérôme, France. Once operational, it will be among the largest in the world, showcasing how this technology is moving beyond pilot projects to full-scale industrial applications. Meanwhile, the U.S. Department of Energy's "Hydrogen Shot" initiative is pushing to lower hydrogen production costs to $1 per kilogram by 2030, a significant drop from the over $3 per kilogram cost recorded in 2022.
This ability to scale production is crucial for creating integrated power systems.
Integration With Power Systems
PEM electrolyzers play a vital role in stabilizing power grids. They absorb excess energy during off-peak times and can adjust output in seconds to align with the availability of renewable energy, addressing the issue of energy intermittency. According to the U.S. Department of Energy:
"Hydrogen fuel and electric power generation could be integrated at a wind farm, allowing flexibility to shift production to best match resource availability with system operational needs and market factors."
When solar and wind energy production dips, the hydrogen stored can either be converted back into electricity using fuel cells or directly used in power generation systems. Additionally, PEM electrolyzers can produce hydrogen at high pressures - up to 34 bar (about 500 psi) - which often eliminates the need for separate mechanical compression equipment.
Such capabilities make PEM technology a key component in creating advanced, integrated energy systems.
Connection to Electrical Equipment
To operate effectively, PEM systems rely on sophisticated power electronics that align with specific voltage and current requirements, typically ranging from 640 V to over 1 kV and 1 kA to 10 kA. Supporting equipment includes transformers, rectifiers, high-pressure pumps, deionized water treatment systems, and heat exchangers. These components must be built to withstand the harsh, acidic environment (pH below 2) of PEM electrolysis, requiring materials like titanium or high-grade stainless steel.
For facilities incorporating PEM systems into existing power infrastructure, Electrical Trader offers specialized components like breakers, transformers, and power distribution tools designed for industrial-scale installations. This ensures compatibility and durability in demanding operational conditions.
Conclusion
PEM electrolysis is reshaping hydrogen production by enabling zero-emission processes when paired with renewable energy sources. Its role in stabilizing power grids that rely on variable solar and wind energy makes it a key player in the shift toward cleaner energy systems.
Since 2001, the capital cost of PEM systems has dropped by over 90%. The U.S. Department of Energy has set an ambitious goal to bring hydrogen production costs down to $1.00 per kilogram by 2031, representing an 80% decrease from current levels. With 43 countries adopting national hydrogen strategies and the U.S. aiming to produce 50 million tonnes of green hydrogen annually by 2050, the industry is scaling up rapidly from pilot programs to massive gigawatt-scale operations. This economic momentum enhances the already impressive technical benefits of PEM systems.
PEM technology stands out for its compact design, ability to deliver high-purity hydrogen, and integrated production pressures, making it highly suitable for industrial use. However, challenges like the reliance on costly platinum and iridium catalysts remain. Fortunately, ongoing research is making strides in addressing these hurdles [5, 18].
By combining hydrogen production with grid stabilization and energy storage, PEM electrolysis supports decarbonization in heavy industries and transportation. As manufacturing scales up and costs continue to decline, this technology is set to play a vital role in developing a resilient and clean energy infrastructure across the United States.
These advancements not only improve cost efficiency but also integrate seamlessly with modern power systems. For professionals exploring cutting-edge hydrogen production and electrical system integration, Electrical Trader provides a trusted source for high-quality electrical components and power distribution solutions.
FAQs
What water quality does a PEM electrolyzer need?
To keep a PEM electrolyzer running efficiently and to extend its lifespan, it needs ultrapure water with minimal impurities. This purity safeguards the membrane and electrodes, preserving their performance and durability over time.
Why are platinum and iridium used in PEM electrolysis?
Platinum and iridium play an essential role in PEM electrolysis due to their outstanding electrocatalytic properties. These characteristics are crucial for efficient water splitting during hydrogen production. Although these materials come with a high price tag, their superior performance ensures the process stays both effective and dependable.
How do PEM electrolyzers connect to grid power safely?
PEM electrolyzers are designed with safety as a top priority, incorporating features like backup systems, fail-safe modes, and remote monitoring to ensure secure grid connections. They adhere to rigorous safety standards and certifications, offering peace of mind when integrated into power systems. Some models are even capable of operating at high pressures without compromising safety. These precautions minimize the risk of failures and help maintain consistent, reliable performance in power generation setups.
